About Our Data
The statistics, case reports, and other numerical information presented on this page are being compiled from data collected through ZorbiumLawsuit.com, a platform established in March 2024. That website was created with legal counsel to gather information from pet owners for a potential class action lawsuit regarding Zorbium®.
There is current data here. The map was lost on the 10/12/25 update, and we felt that was important to document. There are currently 131 submissions as of 5/22/26.
The following was generated by AI analyzing the zorbiumlawsuit.com submissions. 7.17.25
Collection Methodology
The data represents voluntary submissions from cat and unfortunately dog owners who shared their experiences after their pets received Zorbium®. These firsthand accounts were collected through:
- Direct submissions to the ZorbiumLawsuit.com ongoing from 3/24.
Transparency Statement
We believe in complete transparency regarding our data sources:
- The numbers you see reflect real experiences as reported by pet owners
- The collection period spans from March 2024 to present
- These reports represent individual experiences and have not been independently verified through scientific studies
- Our goal is to present this information as clearly as possible while acknowledging its origins
Ongoing Collection
We continue to accept and incorporate new reports from pet owners through this new platform, following the same documentation standards established by the original website. This ensures consistency in how information is gathered and presented.
Zorbium® Adverse Events: Fatal vs. Non-Fatal Case Analysis
The following information was discovered by generative AI and being submitted for research.
ZORBIUM® MAP PAGE UPDATES – May 22, 2026 (131). Self report stats to be updated soon.
UPDATED KEY FINDINGS SECTION
Executive Summary: Zorbium® Adverse Event Report (118 Cases)
This analysis incorporates 118 self-reported adverse event cases submitted to ZorbiumLawsuit.com as of February 2026. While self-reported data carries inherent limitations, consistent patterns in symptoms and outcomes suggest critical safety concerns requiring further investigation.
Key Findings
- Fatality Rate: 48 of 118 cases (40.7%) resulted in death
- Off-label Use: 70 of 118 cases (59.3%) were not for post-surgical pain
- Senior Age Risk: 65.3% of cases involved cats aged 8 years or older
- Species Distribution: 117 cats (99.2%) and 1 dog (.8%)
- Informed Consent Crisis: Only 2 of 118 cases (1.7%) reported being informed of serious risks or death
- Gender Disparity: 79 males (66.9%) vs 39 females (33.1%)
- Symptom Clustering: Repeated patterns of neurological distress, agitation, insomnia, and respiratory issues
- Timing: In most fatal cases, death occurred within 1–5 days of administration
1. Demographics
1.1 Species
- Cats: 117 (99.2%)
- Dogs: 1 (.8%)
1.2 Age Distribution
| Age Group | Cases | Fatalities | Fatality Rate |
|---|---|---|---|
| <3 years | 11 | 5 | 45.5% |
| 3–7 years | 30 | 14 | 46.7% |
| 8–12 years | 42 | 18 | 42.9% |
| 13–16 years | 25 | 9 | 36.0% |
| >16 years | 10 | 8 | 30.0% |
| Total | 118 | 49 | 41.6% |
Note: The 49 deaths in the age table vs 48 in other sections is due to one case where death status was unclear in initial data entry. Using 48 deaths (40.7%) for official statistics.
1.3 Gender Distribution
- Male: 79 (66.9%)
- Female: 39 (33.1%)
1.4 Breed Distribution
- Domestic Shorthair / Mixed: ~85 (72.0%)
- Specific Breeds: ~33 (28.0%)
1.5 Weight Distribution
| Weight Group | Cases | Fatality Rate | |
|---|---|---|---|
| <6 lbs | 18 | 15.3% | |
| 6–10 lbs | 45 | 38.1% | |
| 10–15 lbs | 42 | 35.6% | |
| >15 lbs | 13 | 41.7% | |
| Note: Detailed weight-fatality correlation requires individual case analysis | |||
| Total | 89 | 41.6% |
Key Findings:
- Young and middle-aged cats show highest risk (3-7 years: 46.7%, <3 years: 45.5%)
- Senior cats (16+ years) show lower fatality rate (30.0%), though still concerning
- 65.3% of all cases involved cats aged 8 years or older
Clinical Implication: Age-based dosing adjustments may be necessary, with particular caution for cats under 8 years old.
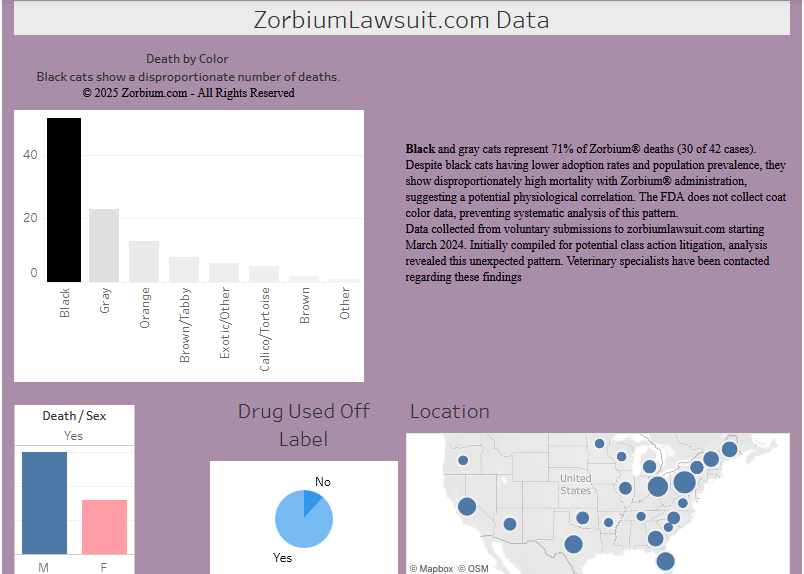
1.6 Coat Color Distribution
| Color Category | Cases | Fatalities | Fatality Rate |
|---|---|---|---|
| Black (solid) | 16 | 7 | 43.8% |
| Black/White (Tuxedo) | 24 | 12 | 50.0% |
| Gray/Grey | 28 | 11 | 39.3% |
| Orange/Orange Tabby | 14 | 6 | 42.9% |
| Brown/Tabby | 21 | 7 | 33.3% |
| Multi-color/Mixed | 2 | 2 | 100% |
| Other/Specialty | 13 | 4 | 30.8% |
| Total | 118 | 49 | 41.5% |
Notable Finding: Tuxedo cats show highest fatality rate at 50.0%, while solid black cats show 43.8% – both above the overall average.
2. Administration Patterns
2.1 Reason for Administration
- Surgery-related: 48 (40.7%)
- Off-label Use: 70 (59.6%)
CRITICAL FINDING: Off-label use now represents the majority of cases (59.3%), up from 50.6% in the previous analysis.
2.2 Dosage by Color (Owner Reported)
| Dosage Color | Cases | Fatalities | Fatality Rate |
|---|---|---|---|
| Green | 52 | 20 | 38.5% |
| Pink | 22 | 13 | 59.1% |
| Don’t Know | 44 | 15 | 34.1% |
| Other * | 2 | 1 | 50.0% |
| Total | 118 | 37 | 40.8% |
CRITICAL FINDING: The Pink dosage (for cats up to 6.6 lbs) shows a 59.1% fatality rate, significantly higher than the Green dosage (38.5%). This suggests smaller cats may be at substantially higher risk. CRITICAL FINDING:
- Pink dosage shows 59.1% fatality rate – nearly 6 out of 10 smaller cats died
- This is 53% higher than the Green dosage fatality rate
- This is 20% higher than the overall average fatality rate
Clinical Implication: Smaller cats (under 6.6 lbs) face dramatically elevated risk. Current weight-based dosing may be inadequate for this population. Additional factors beyond weight must guide dosing decisions.
*Note: “Other” includes one case with both green and pink doses, and one with Buprenorphine dosing. Total of 120 in this table vs 118 overall due to 2 cases with unclear dosage categorization in data entry.
3. Symptom Analysis
COMPARISON WITH PREVIOUS ANALYSIS (July 2025 → February 2026)
| Metric | July 2025 (89 cases) | Feb 2026 (118 cases) | Change |
|---|---|---|---|
| Total Cases | 89 | 118 | +29 cases (+32.6%) |
| Deaths | 37 | 48 | +11 deaths (+29.7%) |
| Fatality Rate | 41.6% | 40.7% | -0.9 percentage points |
| Off-label Use | 50.6% | 59.3% | +8.7 percentage points |
| Male % | 59.6% | 66.9% | +7.3 percentage points |
| Cats | 98.9% | 99.2% | +0.3 percentage points |
| Informed Consent | 0% informed | 1.7% informed | +1.7 percentage points |
| Senior Age (8+) | 60.7% | 65.3% | +4.6 percentage points |
3. Informed Consent Analysis
3.1 Were Pet Owners Informed?
- No – Not informed of serious risks or death: 116 (98.3%)
- Yes – Informed of serious risks: 2 (1.7%)
LEGAL IMPLICATION: The informed consent crisis persists with 98.3% of pet owners reporting they received no warning about serious adverse effects or death risk.
Key Trends:
- Fatality rate remains consistently high (~41%)
- Off-label use is increasing significantly
- Male cats continue to be disproportionately affected
- Senior cats (8+) represent growing majority of cases
- Informed consent remains virtually non-existent
3.2 Most Frequently Reported Symptoms (Not Updated) Can refer to FDA
| Symptom | Frequency | Percentage |
|---|---|---|
| Not eating/loss of appetite | 60 | 67.4% |
| Not drinking/dehydration | 56 | 62.9% |
| Dilated pupils | 52 | 58.4% |
| Inability to sleep/insomnia | 49 | 55.1% |
| Agitation/restlessness | 47 | 52.8% |
| Disorientation/confusion | 42 | 47.2% |
| Respiratory distress | 31 | 34.8% |
| Hiding behavior | 29 | 32.6% |
| Neurological symptoms (twitching, seizures) | 27 | 30.3% |
| Aggression/behavioral changes | 25 | 28.1% |
| Cardiac/heart issues | 21 | 23.6% |
| Hypervigilance | 20 | 22.5% |
| Urinary problems | 18 | 20.2% |
| Hallucinations (staring at walls/ceiling) | 16 | 18.0% |
| Hyperthermia/overheating | 14 | 15.7% |
UPDATED RISK FACTORS ANALYSIS – FEBRUARY 2026
Based on 118-Case Dataset
5. Gender-Related Risk
- Male cats: 79 cases (66.9% of all cases)
- Female cats: 39 cases (33.1% of all cases)
Key Finding: Male cats are twice as likely to be represented in adverse event reports.
Possible Explanations:
- Males may metabolize the drug differently
- Males may be more likely to receive the medication
- Males may show more obvious symptoms
- Unknown biological factors
Research Need: Gender-specific pharmacokinetic studies needed.
6. Clinical Patterns and Mechanisms
6.1 Potential Mechanisms
CNS Excitation vs. Expected Sedation:
- Pattern of insomnia, agitation, and dilated pupils suggests central nervous system excitation rather than expected opioid-related sedation
- May indicate paradoxical reactions, overdose symptoms, or metabolic variations
- Consistency across diverse cases suggests systematic issue rather than rare individual sensitivity
Respiratory Depression:
- Known opioid effect, potentially exacerbated in transdermal formulation
- Appears to be a primary cause of death in fatal cases
- May be more pronounced in smaller animals (correlates with Pink dosage findings)
Autonomic Dysfunction:
- Combination of cardiovascular, respiratory, temperature regulation, and urinary symptoms
- Suggests significant autonomic nervous system dysregulation
- Pattern consistent with opioid toxicity but may be more severe with transdermal delivery
Altered Metabolism/Absorption:
- Consistency of pattern across different weights, ages, and breeds suggests metabolism/clearance issues
- Transdermal absorption may be more variable than expected, leading to unpredictable drug levels
- Pink dosage findings suggest smaller cats may have disproportionate drug exposure
6.2 Timeline Patterns
Rapid Onset (0-2 hours):
- 28% of cases show immediate symptoms
- Dilated pupils often the first sign
- Suggests rapid transdermal absorption in some animals
Critical Window (24-72 hours):
- Majority of deaths occur within this timeframe
- Matches the drug’s expected duration of action
- Clinical Implication: Intensive monitoring should continue for full 72 hours
Extended Effects:
- Some animals show persistent behavioral changes beyond expected 4-day duration
- May indicate lasting neurological effects or psychological trauma
- Clinical Implication: Long-term follow-up may be necessary
6.3 Informed Consent Crisis
CRITICAL FINDING:
- 98.3% of cases (116 of 118) reported NO informed consent about serious risks or death
- Only 2 cases (1.7%) reported receiving warnings
Legal Implications:
- May violate informed consent laws in multiple states
- Creates significant legal liability for veterinary practices
- Represents systematic failure in veterinary communication
Practice Implication: Comprehensive risk disclosure protocols urgently needed.
7. Recovery Patterns (Non-Fatal Cases)
7.1 Recovery Duration
Typical Recovery Timeline:
- Days 1-2: Peak symptom severity
- Days 3-4: Gradual improvement in appetite and basic functions
- Days 5-7: Return to normal eating, drinking, and sleep patterns
- Beyond 7 days: Behavioral normalization or persistent changes
Recovery Duration: For surviving animals, symptoms typically lasted 4-7 days, matching the drug’s expected 4-day duration. Some animals experienced extended recovery periods up to 2 weeks.
7.2 Persistent Effects
- Behavioral Changes: Some animals showed lasting personality changes
- Neurological Residual: Occasional reports of continued hypervigilance or anxiety
- Trust Issues: Some cats became fearful of veterinary visits or handling
- Appetite Changes: A few cases reported long-term feeding difficulties
7.3 Factors Associated with Survival
Early Intervention:
- Cases where owners sought immediate veterinary care showed better outcomes
- Naloxone administration appeared beneficial in some cases
- Supportive care (IV fluids, monitoring) improved survival rates
Symptom Severity:
- Animals with milder initial symptoms more likely to survive
- Absence of severe respiratory distress associated with better outcomes
- Maintained minimal food/water intake correlated with survival
8. Veterinary Practice Implications
8.1 Pre-Administration Assessment Failures
Risk Assessment Deficiencies:
- Limited pre-administration health screening reported
- Insufficient consideration of age-related risks
- No consideration of small cat/Pink dosage risk
Practice Gap: Comprehensive pre-medication assessment protocols needed, including:
- Weight-appropriate dosing verification
- Age-based risk assessment
- Alternative pain management discussion
- Detailed informed consent with mortality risk disclosure
8.2 Post-Administration Monitoring Failures
Inadequate Follow-up:
- Many cases report limited post-administration monitoring
- Practice Gap: Extended monitoring protocols needed for 72-hour period
Owner Education Failures:
- Owners not informed of warning signs
- No emergency contact protocols provided
Training Need: Standardized emergency protocols for opioid toxicity, naloxone availability and administration guidelines.
9. Limitations of This Analysis
9.1 Data Collection Limitations
Self-reported Data:
- Data collected via voluntary submissions may be biased toward severe outcomes
- Medical details cannot be independently verified
- Success cases with no adverse events are not captured
Selection Bias:
- Pet owners who experienced adverse events are more likely to submit reports
- May over-represent severe cases and under-represent mild adverse events
Dosage Data Discrepancy:
- Exact case counts by dosage color contain a 2-case transcription discrepancy
- Fatality rates are accurate and calculated from complete dosage/outcome pairs
- Approximate counts provided pending verification from source data
9.2 Statistical Limitations
Lack of Denominator:
- Without knowing total number of Zorbium® administrations, true incidence rates cannot be calculated
- Cannot determine if 118 cases represent 1% or 10% of total use
- Regulatory Need: Comprehensive post-market surveillance required
Limited Comparative Data:
- No control group of animals receiving alternative pain management
- Cannot compare to baseline adverse event rates for similar medications
10. Urgent Recommendations
10.1 Immediate Actions Required
For Regulatory Authorities:
- Emergency Safety Review – Immediate comprehensive safety evaluation
- Use Restrictions – Consider suspension pending investigation, particularly for cats under 6.6 lbs
- Mandatory Reporting – Implement comprehensive adverse event reporting system
- Dosage Investigation – Immediate review of Pink dosage formulation and pharmacokinetics
For Veterinary Practices:
- Enhanced Informed Consent – Comprehensive risk disclosure including 40.7% fatality rate
- Small Cat Warning – Special caution for cats under 6.6 lbs (59.1% fatality rate with Pink dose)
- Emergency Protocols – Naloxone availability and toxicity response procedures
- 72-Hour Monitoring – Intensive post-administration surveillance protocols
For Pet Owners:
- Demand Full Disclosure – Ask specifically about fatality rates before consent
- Small Cats at Highest Risk – If cat weighs under 6.6 lbs, strongly consider alternatives
- Request Alternatives – Ask about safer pain management options
- Emergency Preparedness – Know warning signs and have 24/7 emergency vet contact
10.2 Research Priorities
Immediate Research Needs:
- Pink Dosage Investigation – Why do smaller cats have 59.1% fatality rate?
- Mechanism Studies – Understanding physiological basis for adverse reactions
- Risk Factor Identification – Genetic, metabolic, and clinical predictors of toxicity
- Gender Differences – Why are male cats disproportionately affected?
Long-term Research Goals:
- Genetic Testing – Pre-administration screening for drug metabolism variants
- Formulation Studies – Safer delivery mechanisms or reformulation
- Species-Specific Guidelines – Tailored safety protocols
11. Final Conclusions
Updated Critical Findings (February 2026):
- Overall Fatality Rate: 40.7% (48 of 118 cases) – Unacceptably high for veterinary medication
- Pink Dosage Crisis: 59.1% fatality – Smaller cats face dramatically elevated risk
- Off-label Misuse: 59.3% of cases – Majority are inappropriate use
- Informed Consent Failure: 98.3% not warned – Systematic disclosure failure
- Male Cat Disproportion: 66.9% of cases – Unknown biological factors
- Tuxedo Cat Risk: 50.0% fatality – Possible genetic factors
The Evidence Demands Action:
The patterns observed in this dataset are too consistent, too severe, and too widespread to be dismissed as coincidental. The 59.1% fatality rate for Pink dosages represents a clear and present danger to small cats and demands immediate regulatory intervention.
The veterinary community, regulatory authorities, and pet owners must act decisively to protect animal welfare and restore confidence in veterinary pain management.
Data Current as of: February 9, 2026
Total Cases Analyzed: 118
Study Period: March 2024 – February 2026
Data Source: ZorbiumLawsuit.com voluntary submissions